Alex Chelton, OMS 2027; Ethan Lawson, OMS 2027; Saavan Boghara, OMS 2026; and Francis E. Jenney Jr., PhD
Philadelphia College of Osteopathic Medicine – Georgia Campus
DOI: http://doi.org/10.70709/ellc5ccpa22
Abstract
Background
GLP-1 receptor agonist medication usage has seen a rapid increase in usage not only for diabetic patients but also for obese and overweight patients who are searching for alternative weight loss methods. Although these medications are proving useful in helping patients lose weight, there are notable side effects that patients should consider.
Methods
A retrospective case-control study was conducted using data from the National Institutes of Health’s All of Us Database. Patients were included based on the diagnosis of overweight and obesity. Patients were excluded based on a diagnosis of Type 1 or Type 2 Diabetes Mellitus, an A1C level greater than 6.5, and a diagnosis of conditions that would increase the risk of fragility fractures.
Results
After including 12,171 patients and analyzing fracture risk in the GLP-1 RA exposure group, we found the relative risk to be 0.69 and the odds ratio for fracture risk in the GLP-1 RA exposure group to be 0.67.
Conclusion
Our findings suggest that GLP-1 RAs may be protective against fractures in patients.
Keywords: GLP-1, obesity, overweight, fractures, bone, pharmacotherapy
Introduction
GLP-1 RAs were first identified in the 1980s and received FDA approval in 2005 as anti-hyperglycemic agents for the treatment of diabetes (1). As incretin hormones, GLP-1 RAs stimulate insulin secretion, suppress glucagon release, and reduce peak plasma glucose concentrations (1, 2). Over time, their therapeutic indications have expanded to include cardiovascular protection, renal protection through the microvascular benefits of tight glycemic control, and weight loss via appetite suppression (3).
The potential impact of GLP-1 RAs on bone fracture risk warrants further investigation. Many individuals prescribed these agents have T2DM, a known independent risk factor for fractures (4). In contrast, these patients often present with higher BMI, which is generally associated with a reduced fracture risk (5). Complicating matters, GLP-1 RA–induced weight loss has itself been linked to an increased risk of fractures (6). Current literature on the relationship between GLP-1 RA use and fracture risk is inconsistent, with studies reporting neutral, protective, or harmful effects (7). The present study aims to clarify this association by examining fracture risk in patients using GLP-1 RAs who have no known comorbidities that independently influence fracture risk.
Materials and Methods
A retrospective case-controlled study was performed using data from the NIH’s AllofUs Database. Patients were included based on the NIH Concept ID diagnosis of obesity assigned by the database. Exclusion criteria included the Concept ID for diagnoses of Type 1 Diabetes Mellitus, Type 2 Diabetes Mellitus, Elevated HbA1C levels (≥6.5), and conditions that would increase fracture risk, including diagnoses for: Osteoporosis, Rheumatoid Arthritis, Chronic Kidney Disease, and Long-term Corticosteroid Use. After creating our cohort from the approximately 849,000 patients in the AllofUs database, relative risks and odds ratios were calculated using the 12,171 patients who met the inclusion criteria for our study.

Results
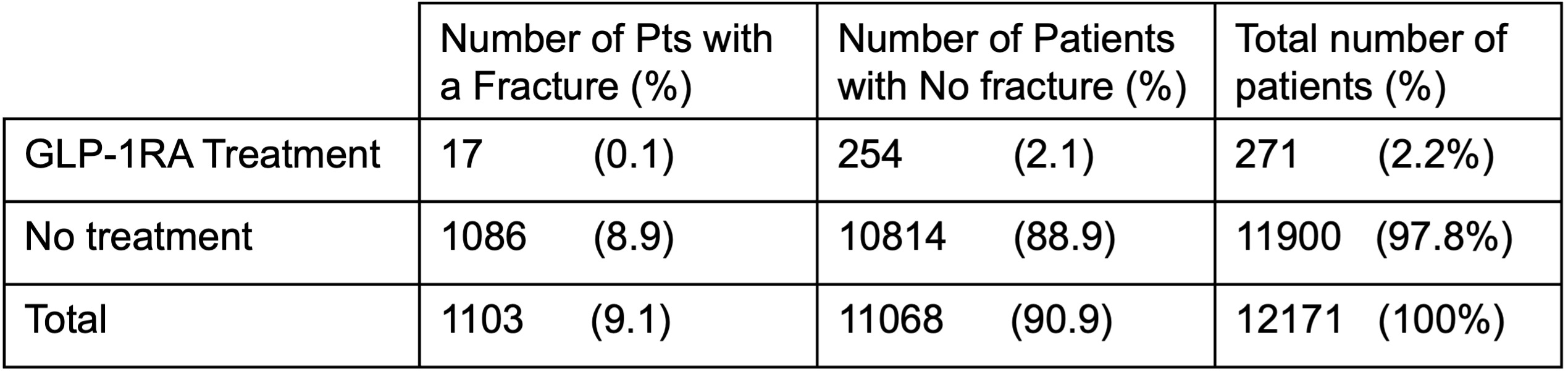
Our study included 12,171 patients who met the inclusion criteria. Two hundred seventy-one patients from our sample of 12,171 had a history of being prescribed GLP-1 RA for weight loss purposes, representing 2.2% of our total included patient population. Following the exposure to GLP-1 RA, 17 of the 271 patients exhibited a history of at least one fracture following the usage of GLP-1 RA medications, which is 0.13% of the total included population and 6.3% of the GLP-1 RA exposed patients. The remaining 11,900 patients who met the inclusion criteria had no history of being prescribed GLP-1 RA. Within this group, 1,086 patients had a history of fracture, representing 8.9% of the total included patient population. The remaining 10,814 patients did not have a history of fracture, which represented the majority of our included patients, at 88.9% of the total included patients. Using these numbers, we were able to calculate that the Relative Risk of GLP-1 RA exposure was 0.69 and the Odds Ratio of GLP-1 RA exposure was 0.67. The data in Figure 2 compares the total number of fractures in patients taking GLP-1RAs with that of the larger population that did not receive the treatment.
Discussion
With a Relative Risk Ratio of 0.67 and an Odds Ratio of 0.69, our findings suggest that GLP-1 RA exposure could potentially provide a protective effect against fractures, with our patients having been exposed to GLP-1 RA having lower odds of experiencing a fracture when compared to the unexposed group. This finding is consistent with studies that have demonstrated that some GLP-1 RAs, like Liraglutide, may be associated with modest reductions in fracture risks in patients with T2DM who have been prescribed a GLP-1 RA for at least one year (7, 8, 9).
The mechanisms responsible for the potential protective effect on fracture risk in patients involve both direct and indirect mechanisms. Some studies have linked GLP-1 RA to increased bone formation and inhibition of bone resorption by modulating the functionality of osteoblasts and osteoclasts, respectively. More specifically, GLP-1 RAs have been linked to increased bone formation markers, such as P1NP, osteocalcin, and bone-specific alkaline phosphatase, while simultaneously reducing bone resorption markers, like β-CTX (10, 11, 12). Mechanistically, GLP-1 RA activation in bone cells has been shown to suppress osteoclastogenesis by inhibiting the NF-κB and MAPK signaling pathways, reducing bone resorption (13). There is also evidence to support that GLP-1 RAs favor bone formation by inhibiting RANKL-mediated osteoclast activation secondary to increasing OPG expression while simultaneously promoting osteogenic differentiation of bone mesenchymal stem cells and decreasing sclerostin expression, which in turn enhances Wnt/-catenin signaling, leading to bone formation (14).
While these mechanisms can explain the protective effect that GLP-1 RA exposure had on the fracture risk of the exposed patients in our study, it is important to consider the fact that the findings mentioned were from studies primarily conducted on patients prescribed GLP-1 RA who had a diagnosis of T2DM, as this is what the drugs were originally FDA-approved to treat. When exploring any potential differences between the mechanisms of bone metabolism in patients with and without T2DM, we found that patients with T2DM typically have lower levels of bone formation markers (i.e. osteocalcin and P1NP and bone resorption markers (C-terminal telopeptide of type 1 collagen), leading to decreased levels of bone turnover (15, 16, 17, 18).
While patients with T2DM may have lower levels of bone turnover markers, the markers responsible for bone turnover are the same as those in patients without T2DM. A reasonable assumption can be made that the mechanisms identified as providing a protective effect against fractures in patients with T2DM are likely to have a similar protective effect in our sample of patients without T2DM. Additionally, it is reasonable to consider that the decreased fracture risk in the exposure group of our study is multifactorial, with factors including weight loss resulting from being prescribed GLP-1 RA and increased motivation to adopt a healthier lifestyle. More specifically, we hypothesize that the patient population, including those pursuing pharmacological assistance to aid their weight loss journey, may disproportionately represent the more motivated patients. These more motivated patients may exhibit other positive behaviors such as maintaining a healthy diet and engaging in regularexercise, which could provide further protection against fractures.
Our study was subject to limitations resulting from the sample size that met our inclusion criteria, as well as restrictions that we faced with the AllofUs Database regarding exporting patient data to allow for propensity matching our patients. Using GLP-1 RAs for weight loss control is a relatively new tool for providers to apply to patient care, with Liraglutide and Semaglutide obtaining FDA approval for weight management in non-diabetic populations in 2014 and 2021, respectively (19, 20, 21). GLP-1 receptor agonists gained significant increases in prescriber popularity in 2021, with Semaglutide accounting for 60% of prescriptions by 2023 due to its superior efficacy in clinical trials compared to Liraglutide (22). Given the recent rise in the use of these medications, we were limited in the ability to analyze any potential side effects of using these medications for weight loss, including their impact on a patient’s fracture risk, that may present outside of the 10 years of data available to us, especially with a majority of the data coming since the spike in popularity since 2021. In addition, our study was limited by the inability to propensity match patients who met the inclusion criteria, thereby eliminating potential confounding variables, such as age, sex, BMI, and other lifestyle factors.
In future studies, we hope to use more updated data as the population of patients using these medications for weight loss increases. Once more data is available, it will be possible to propensity match patients and control for confounding variables that may be impacting fracture risk in these patients. This expanded dataset will also enable more representative and predictive statistics for patients using GLP-1 RAs for weight loss. Aside from short-term outlooks, these medications need to be studied for long-term effects in these patients to determine if there are any lasting implications that have yet surfaced.
Conclusions
GLP-1 RA medications have become a very popular method to achieve weight loss in overweight and obese patients. While patients flock to new methods of losing weight, clinicians must maintain skepticism and ensure safety for their patients. Although this study doesn’t directly demonstrate GLP-1 RA medications are safe or protective for fractures, these findings do show a decreased risk of fracture amongst this patient population. This finding could be due to the weight loss effects of these medications, thus reducing the force on patients’ bones. Aside from weight loss, many patients experience an increase in their activity level on these medications as they adopt new habits to sustain their weight loss, which can, in turn, enhance bone strength. These effects, combined with a decrease in BMI, can lead to a reduction in fracture risk. While other variables may affect or account for these results, the potential role of GLP-1 RA use in fracture risk requires further study. These findings are significant as this expanding patient population begins to increase usage of these medications. It is critical to understand the implications of novel medications later in life.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- Yao H, Zhang A, Li D, et al. Comparative effectiveness of GLP-1 receptor agonists on glycaemic control, body weight, and lipid profile for type 2 diabetes: systematic review and network meta-analysis. BMJ. 2024;384:e076410. Published 2024 Jan 29. doi:10.1136/bmj-2023-076410
- Collins L, Costello RA. Glucagon-Like Peptide-1 Receptor Agonists. In: StatPearls. Treasure Island (FL): StatPearls Publishing; February 29, 2024.
- Zhao X, Wang M, Wen Z, et al. GLP-1 Receptor Agonists: Beyond Their Pancreatic Effects. Front Endocrinol (Lausanne). 2021;12:721135. Published 2021 Aug 23. doi:10.3389/fendo.2021.721135
- Li GF, Zhao PP, Xiao WJ, Karasik D, Xu YJ, Zheng HF. The paradox of bone mineral density and fracture risk in type 2 diabetes. Endocrine. 2024;85(3):1100-1103. doi:10.1007/s12020-024-03926-w
- Cherukuri L, Kinninger A, Birudaraju D, et al. Effect of body mass index on bone mineral density is age-specific. Nutr Metab Cardiovasc Dis. 2021;31(6):1767-1773. doi:10.1016/j.numecd.2021.02.027
- Jiang BC, Villareal DT. Weight Loss-Induced Reduction of Bone Mineral Density in Older Adults with Obesity. J Nutr Gerontol Geriatr. 2019;38(1):100-114. doi:10.1080/21551197.2018.1564721
- Zhang Y, Chen G, Wang W, Yang D, Zhu D, Jing Y. Association of Glucagon-like peptide-1 receptor agonists use with fracture risk in type 2 diabetes: A meta-analysis of randomized controlled trials. Bone. 2025;192:117338. doi:10.1016/j.bone.2024.117338
- Cheng L, Hu Y, Li YY, et al. Glucagon-like peptide-1 receptor agonists and risk of bone fracture in patients with type 2 diabetes: A meta-analysis of randomized controlled trials. Diabetes Metab Res Rev. 2019;35(7):e3168. doi:10.1002/dmrr.3168
- Zhang YS, Weng WY, Xie BC, et al. Glucagon-like peptide-1 receptor agonists and fracture risk: a network meta-analysis of randomized clinical trials. Osteoporos Int. 2018;29(12):2639-2644. doi:10.1007/s00198-018-4649-8
- Tan Y, Liu S, Tang Q. Effect of GLP-1 receptor agonists on bone mineral density, bone metabolism markers, and fracture risk in type 2 diabetes: a systematic review and meta-analysis. Acta Diabetol. 2025;62(5):589-606. doi:10.1007/s00592-025-02468-5
- Alalwani YJ, Alshahrani AS, Alsudays AI, et al. Differential effects of GLP-1 receptor agonists on diabetic osteopathy in type 2 diabetes: a patient-stratified network meta-analysis. BMC Musculoskelet Disord. 2025;26(1):742. Published 2025 Aug 1. doi:10.1186/s12891-025-09022-y
- Li X, Li Y, Lei C. Effects of Glucagon-Like Peptide-1 Receptor Agonists on Bone Metabolism in Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis. Int J Endocrinol. 2024;2024:1785321. Published 2024 Sep 14. doi:10.1155/2024/1785321
- Li Z, Li S, Wang N, Xue P, Li Y. Liraglutide, a glucagon-like peptide-1 receptor agonist, suppresses osteoclastogenesis through the inhibition of NF-κB and MAPK pathways via GLP-1R. Biomed Pharmacother. 2020;130:110523. doi:10.1016/j.biopha.2020.110523
- Luo G, Liu H, Lu H. Glucagon-like peptide-1(GLP-1) receptor agonists: potential to reduce fracture risk in diabetic patients?. Br J Clin Pharmacol. 2016;81(1):78-88. doi:10.1111/bcp.12777
- Yang J, Zhang Y, Liu X, Chen B, Lei L. Effect of type 2 diabetes on biochemical markers of bone metabolism: a meta-analysis. Front Physiol. 2024;15:1330171. Published 2024 Jul 19. doi:10.3389/fphys.2024.1330171
- Hygum K, Starup-Linde J, Harsløf T, Vestergaard P, Langdahl BL. MECHANISMS IN ENDOCRINOLOGY: Diabetes mellitus, a state of low bone turnover – a systematic review and meta-analysis. Eur J Endocrinol. 2017;176(3):R137-R157. doi:10.1530/EJE-16-0652
- Starup-Linde J, Lykkeboe S, Handberg A, et al. Glucose variability and low bone turnover in people with type 2 diabetes. Bone. 2021;153:116159. doi:10.1016/j.bone.2021.116159
- Starup-Linde J. Diabetes, biochemical markers of bone turnover, diabetes control, and bone. Front Endocrinol (Lausanne). 2013;4:21. Published 2013 Mar 8. doi:10.3389/fendo.2013.00021
- Elmaleh-Sachs A, Schwartz JL, Bramante CT, Nicklas JM, Gudzune KA, Jay M. Obesity Management in Adults: A Review. JAMA. 2023;330(20):2000-2015. doi:10.1001/jama.2023.19897
- Gudzune KA, Kushner RF. Medications for Obesity: A Review. JAMA. 2024;332(7):571-584. doi:10.1001/jama.2024.10816
- Wharton S, Blevins T, Connery L, et al. Daily Oral GLP-1 Receptor Agonist Orforglipron for Adults with Obesity. N Engl J Med. 2023;389(10):877-888. doi:10.1056/NEJMoa2302392
- Ukhanova M, Wozny JS, Truong CN, Ghosh L, Krause TM. Trends in glucagon-like peptide 1 receptor agonist prescribing patterns. Am J Manag Care. 2025;31(8):e228-e234. Published 2025 Aug 1. doi:10.37765/ajmc.2025.89778


