Mitchell Rentschler, OMS-IV1; Mason Kyle, OMS-IV1; John Ashurst, D.O.2; Amanda Rivera-Begeman, D.O.3; Benjamin Walters, D.O.4
1Arizona College of Osteopathic Medicine
2Department of Assessment Midwestern University – Arizona College of Osteopathic Medicine
3Department of Pathology, Sanford School of Medicine and Department of Pathology, Avera McKennan Hospital and University Health Center
4Department of Surgery, Sanford School of Medicine and Department of Orthopedics, Avera McKennan Hospital and University Health Center
DOI: http://doi.org/10.70709/i15fg7w8t4
Abstract
Case
A 42-year-old woman with a history of alcoholic liver disease presented with progressive left hip pain over three months. Imaging revealed instability of the superior left femoral head with cortical breakthrough and a well-circumscribed mixed sclerotic and cystic lesion in the left femoral neck. The patient was diagnosed with a liposclerosing myxofibrous tumor and avascular necrosis of the femoral head. She underwent a left total hip arthroplasty with cerclage wire and returned to work with significant pain relief.
Conclusion
Liposclerosing myxofibrous tumors are rare, benign fibro-osseous tumors usually diagnosed incidentally and have not been previously reported to be associated with the development of avascular necrosis. Factors like corticosteroid use, alcohol abuse, and possibly liposclerosing myxofibrous tumors can impair blood flow and contribute to avascular necrosis. Patients with liposclerosing myxofibrous tumors, particularly those with additional risk factors, should be monitored closely for avascular necrosis development.
Keywords: Liposclerosing myxofibrous tumor, avascular necrosis, benign femur lesion, case report
Introduction
Liposclerosing myxofibrous tumor (LSMFT) is a rare, benign tumor first described by Ragsdale and Sweet in 1986 1-5. LSMFT are typically discovered incidentally on radiograph, computed tomography (CT), or magnetic resonance imaging (MRI) with similar prevalence among males and females 2,3,6,7. The true prevalence of LSMFT has not been established due to the absence of population-based studies; however, because these tumors are often incidental and asymptomatic, the actual incidence is likely underestimated. Microscopic analysis can reveal a variety of histopathologic features, including myxofibrous tissue, fibrous dysplasia-like features, and ischemic ossification with an unclear pathogenesis leading to debates about whether LSMFT constitutes a distinct diagnosis versus a variant of fibrous dysplasia or intraosseous lipoma 3,6,8,9. Most LSMFT occur in the proximal femur, with occasional reports in the distal femur and cranial vault, and complications from these tumors are rare, except for infrequent malignant transformations 1,3,4,6,7,9. In this case report, the authors document a patient diagnosed with LSMFT who initially presented with avascular necrosis (AVN). To the authors knowledge this is the first report linking LSMFT to AVN, and we will discuss the clinical presentation, imaging, diagnosis, and management of this case.
Statement of Informed Consent
All information in this case report has been deidentified and the patient gave informed written consent for publication of the data.
Case Report
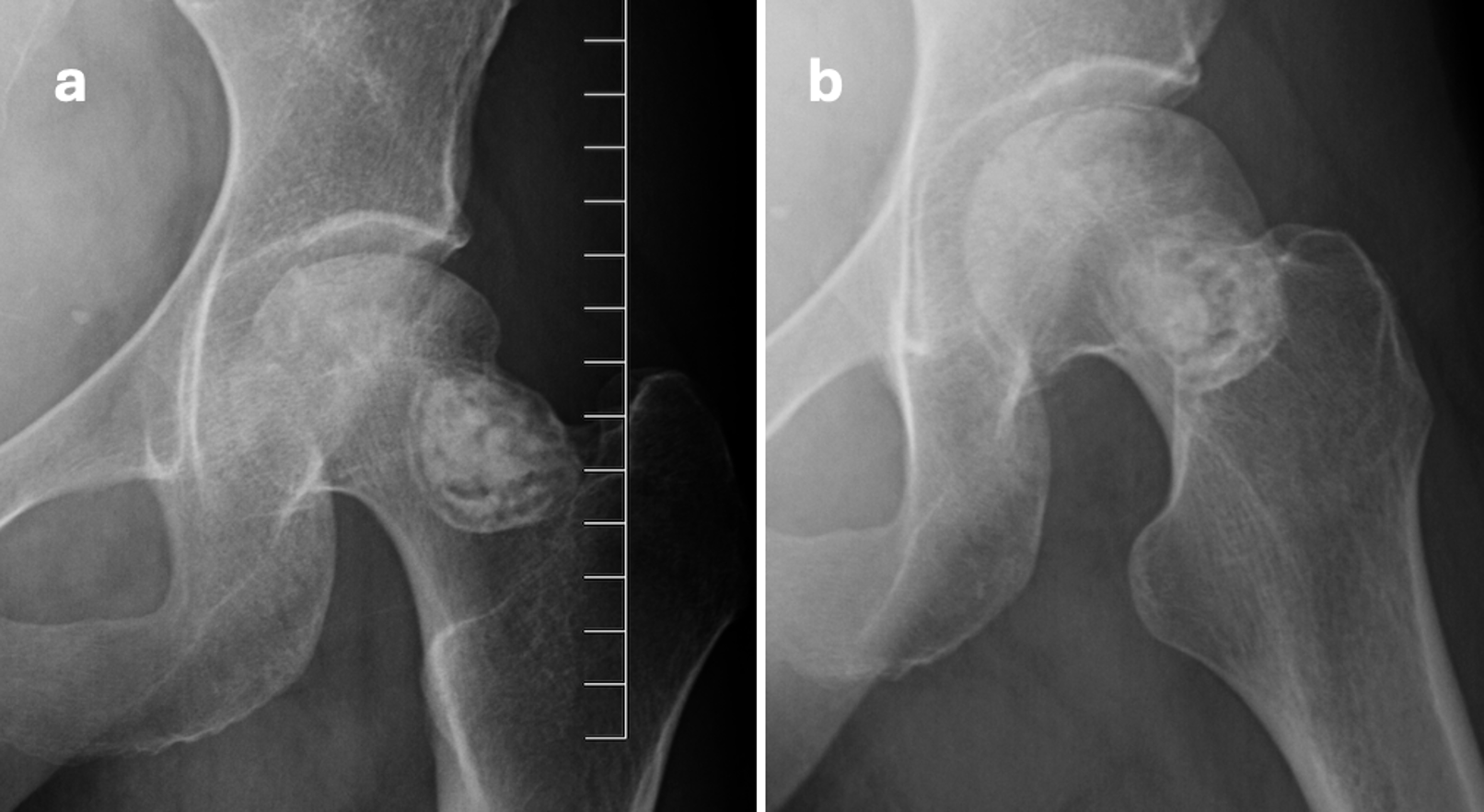
A 42-year-old woman presented with worsening left hip pain over the past three months, without any history of trauma. She reported pain primarily in the anterior hip and groin, exacerbated by rotation. Over-the-counter anti-inflammatories provided minimal relief, leading to a prescription for tramadol. Her past social history included alcohol and tobacco abuse, with sustained sobriety for over one year, as well as insomnia, anxiety, and depression. The patient’s medical history comprised a Weber B fibula fracture, gastrointestinal bleeding, an abnormal cervical Pap smear, alcoholic liver damage, ascites, and jaundice. Current medications included acamprosate, cholecalciferol, furosemide, gabapentin, magnesium chloride, meloxicam, multivitamins, potassium chloride, single prednisone taper completed two months prior to presentation, spironolactone, tramadol, trazodone, and vitamin B complex. On musculoskeletal examination, the left hip exhibited decreased functional active and passive range of motion (ROM) with pain at the endpoints. Pain intensified with internal and external rotation, accompanied by mild crepitus. There was a positive flexion abduction external rotation (FABER) test and log roll test of the left hip. Radiograph of the left hip revealed a 3.6 cm sclerotic lesion in the left femoral neck, with lucency and cortical irregularity apparent in the superior left femoral head (Figure 1a and 1b).
MRI with T1- and proton density (PD)-weighted sequences and gadolinium contrast demonstrated AVN of the superior femoral head with instability and cortical breakthrough (Figure 2a and 2b).
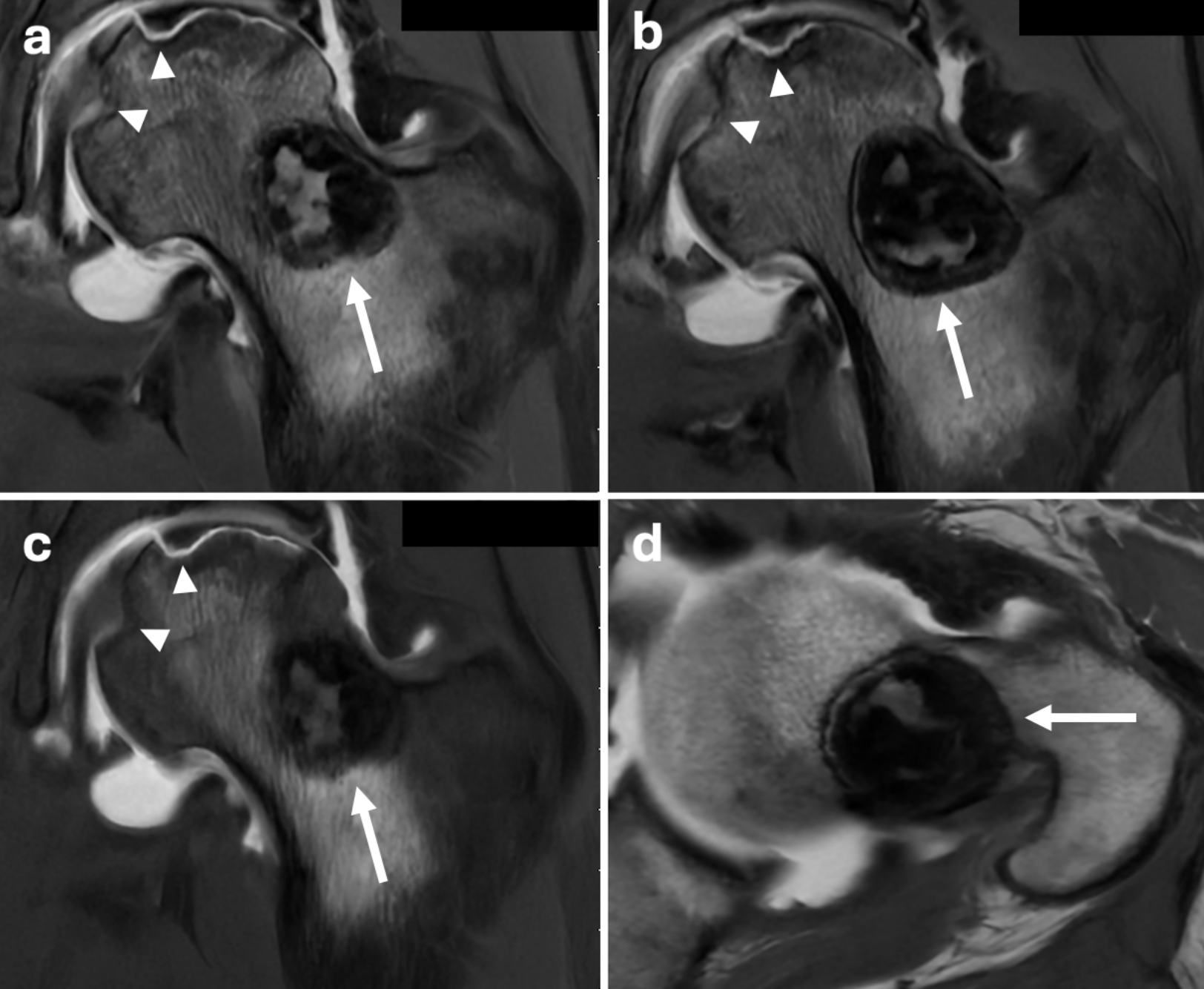
Additionally, a well-circumscribed mixed sclerotic and cystic lesion measuring 2.4 x 2.6 x 2.5 cm in the femoral neck, without aggressive features, was consistent with LSMFT (Figure 2c and 2d).
The patient underwent cementless left total hip arthroplasty. A posterior approach was used, with capsulotomy made directly over the posterior greater trochanter down to the femoral neck, ensuring surgical cuts remained below the LSMFT. The femoral head displayed typical early AVN findings, including cartilage loss and sclerotic bone on the lateral distal neck. Although there was no evidence of fracture around the medial calcar, a prophylactic cerclage cable was placed proximal to the lesser trochanter due to the LSMFT. The hip was reduced, achieving satisfactory ROM and leg lengths, followed by extensive irrigation and capsular closure. The patient was transferred to the post-anesthesia care unit without any complications and was discharged the following day in good condition.
Histological analysis demonstrated fraying patches of articular cartilage with underlying trabecular bone exhibiting osteocyte nuclear dropout consistent with AVN. Examination of the sclerotic lesion revealed fragments of well-demarcated, variably mineralized, and sclerotic bone with interspersed clusters of foamy macrophages and a few lobules of mature adipocytes to suggest a benign sclerotic bone tumor (Figure 3a, 3b, and 3c).
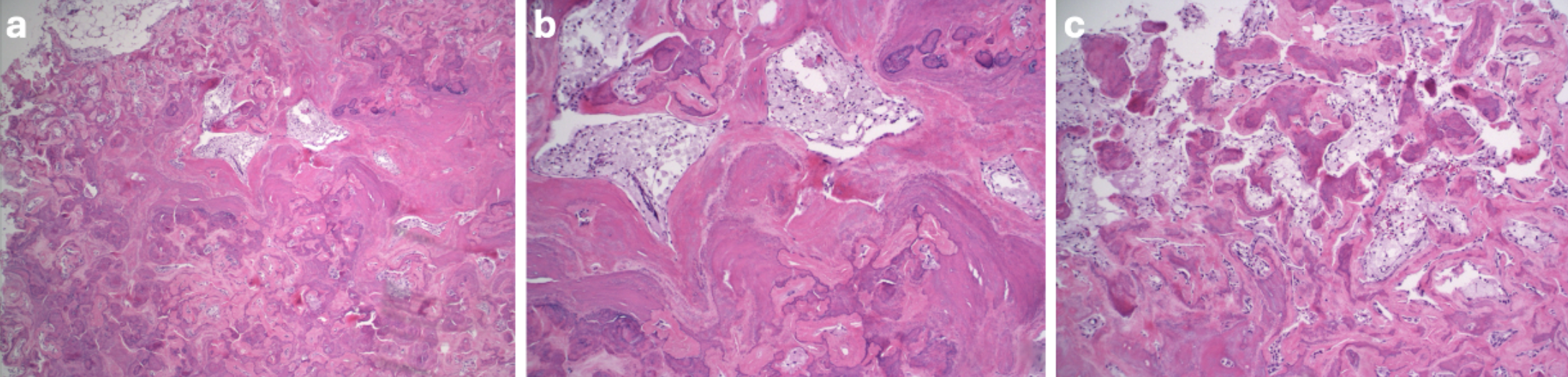
No distinct osteoblastic rimming or fibrous component was appreciated helping to rule out other diagnoses.
The patient began physical therapy shortly after surgery and experienced an uncomplicated recovery. Follow-up appointments and imaging at two and six weeks post-operatively were unremarkable (Figure 4a and 4b).
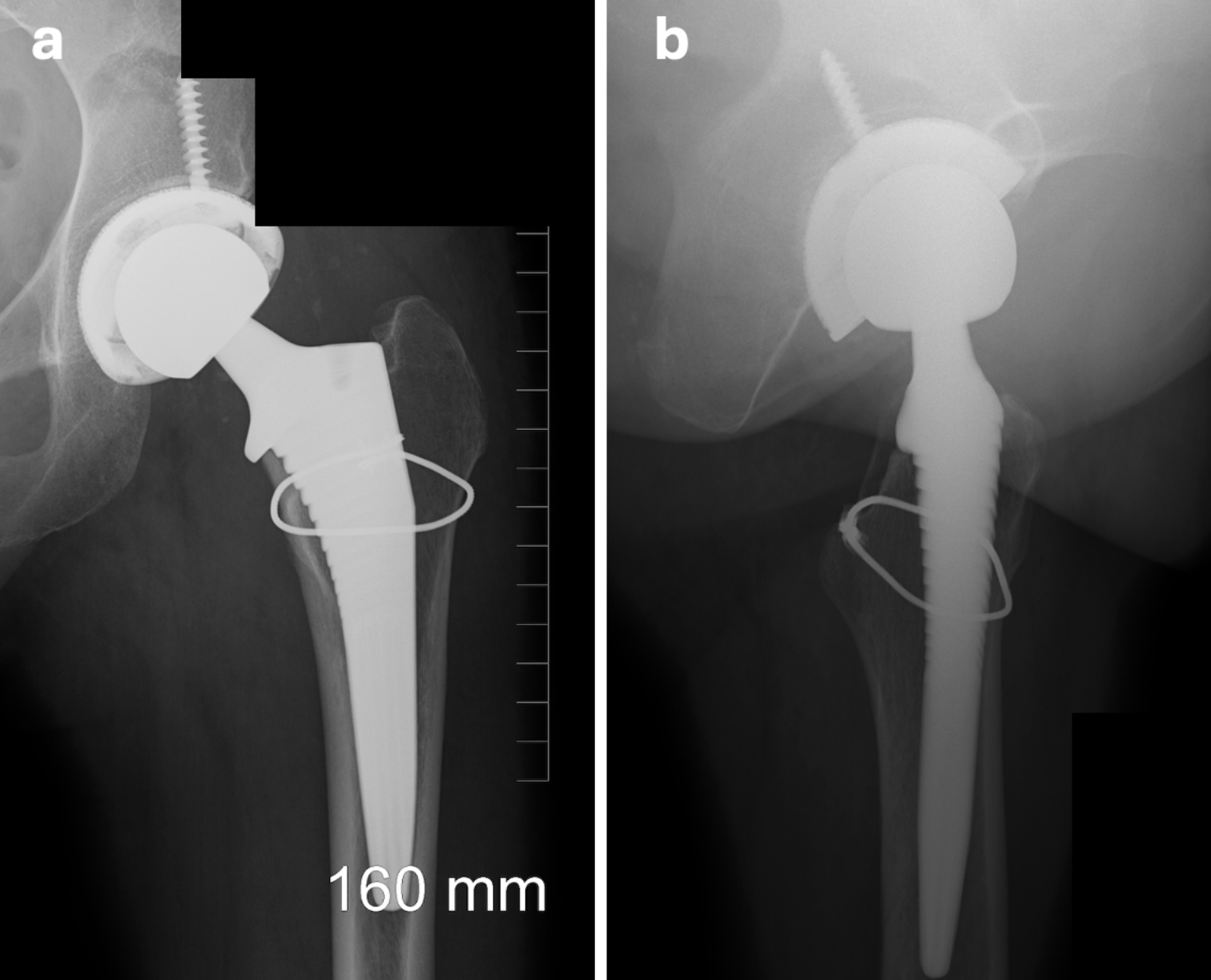
She reported significant pain relief and was able to return to work without restrictions. At one-year follow-up, the patient remained pain-free in the left hip, with imaging demonstrating no abnormalities (Figure 5a and 5b).
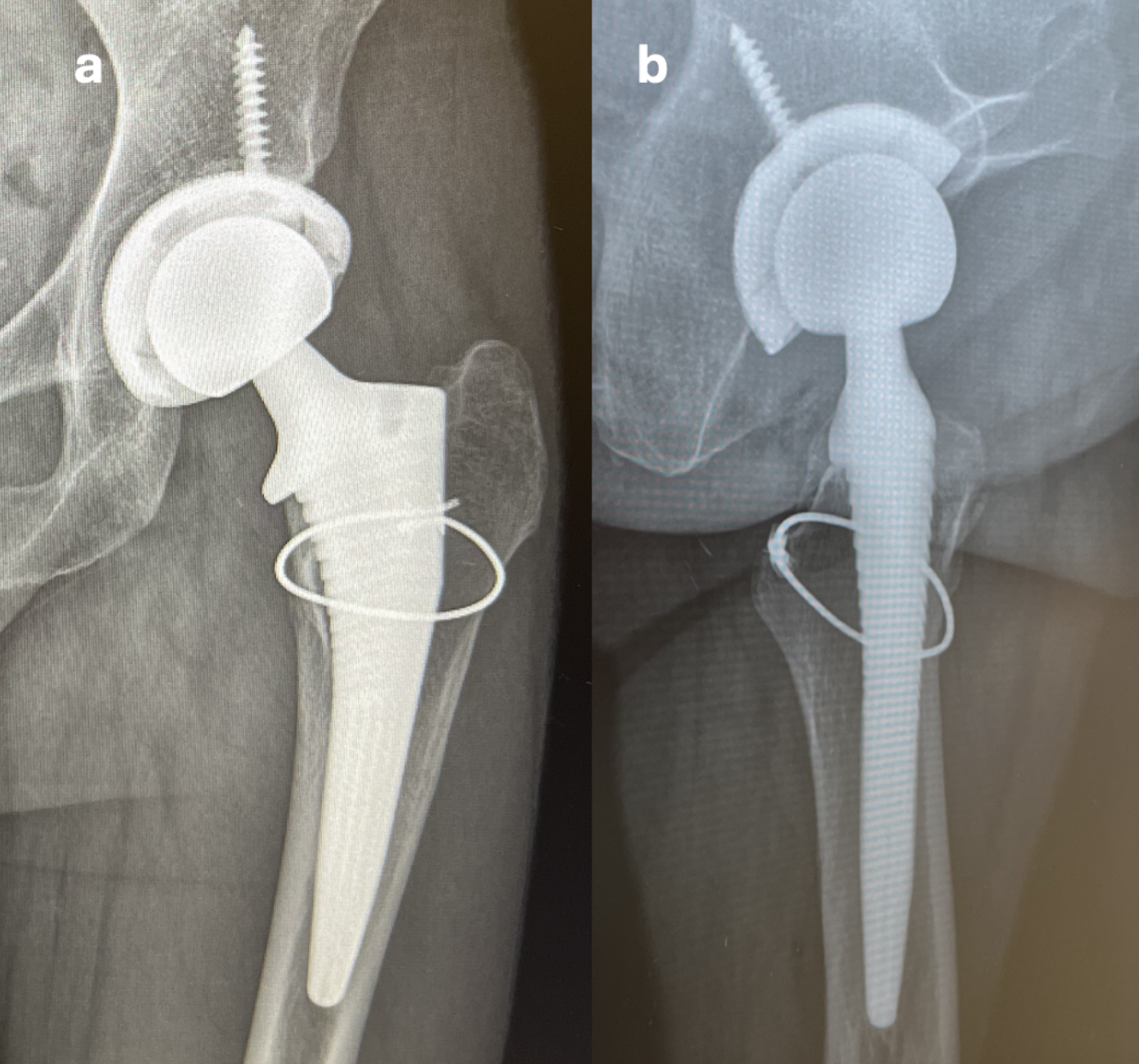
Throughout the year, she has continued working full-time, participated in regular physical activity, and expressed high satisfaction with her functional outcome and overall quality of life.
Discussion
In 1986, Ragsdale and Sweet described the first case of LSMFT 1-5. LSMFT are classified as a rare, benign tumors with approximately 80% to 90% of documented cases located in the intertrochanteric region 1,3. The age of onset ranges from 15 to 80 years, with a typical presentation around 40 years, and the incidence is equal among males and females 2,3,6,7. Most LSMFT are discovered incidentally via radiograph or MRI 3. Radiographs typically reveal an indolent growth of a lytic lesion with sclerotic margins, while MRI T1-weighted images show moderate homogeneity and high-intensity heterogeneity on T2-weighted images 6,9. Additionally, osteoblastic rimming is a common finding in osteofibrous dysplasia and occasionally fibrous dysplasia helping to rule out these diagnoses based on imaging 3.
The histopathologic diversity makes the diagnosis of LSMFT challenging, as histological examination often reveals a mixture of lipoma, myxoma, myxofibroma, fibroxanthoma, fat necrosis, and fibrous dysplasia-like features 3,5,8. Due to variations in histological findings, some researchers argue that LSMFT is not a distinct entity but rather a variant of fibrous dysplasia or interosseous lipomas 8,10,11.
Despite being classified as benign, LSMFT carries a reported malignant transformation rate of up to 16%, along with distinct histological features, making it an important clinical entity 3,4,7. The main recognized complications of LSMFT include malignant transformation, bone pain, and pathological fracture 3. However, this case introduces a previously unidentified complication: AVN. In the United States, the annual incidence of AVN is estimated at 20,000 to 30,000 cases, typically affecting individuals aged 20 to 40 12. The most common causes of AVN of the femoral head are hip fractures, dislocations, corticosteroids, and alcohol abuse 13,14. Clinically, AVN presents as hip pain that radiates to the groin, stiffness, and decreased ROM; it can be diagnosed based on clinical findings and X-ray imaging, although MRI remains the gold standard 13.
The pathogenesis of non-traumatic AVN of the femoral head is underdetermined, however, it is definitively linked to reduced blood flow 14. The proposed mechanisms by which corticosteroids and alcohol induce ischemia involve adipocyte hyperplasia, increased intra-osseous pressure, endothelial damage, and subsequent reduction in blood flow 13,14. In our patient, a history of corticosteroid use and prior alcohol abuse, despite one year of sobriety, could have contributed to the development of AVN, potentially independent of the LSMFT. However, corticosteroid-induced AVN is typically dose- and duration-dependent, and given the short-term tapering course of prednisone in our patient, this etiology is less likely 15. Similarly, alcohol-induced AVN usually presents within one year of heavy alcohol consumption 16. Although AVN may have developed independently of the LSMFT, its presence in the ipsilateral femur suggests a potential contributory role. Corticosteroid and alcohol effects on ischemia may be exacerbated by a growing LSMFT, which, despite its benign nature, can raise intraosseous pressure and impair femoral head perfusion. The etiology of AVN in our patient is likely multifactorial. Treatment for asymptomatic AVN of the femoral head and LSMFT is usually conservative while arthroplasty is reserved for symptomatic patients 3,12.
Conclusion
LSMFT are rare, benign tumors associated with several previously reported complications, including malignant transformation, bone pain, and pathological fractures. However, LSMFT may increase the risk of AVN of the femoral head due to lesion growth which ultimately reduces blood flow. Patients diagnosed with LSMFT should undergo heightened monitoring either clinically or with imaging for AVN, particularly when presenting with high-risk factors such as corticosteroid use or alcohol abuse.
References
- Zhang M, Zhang D, Yu W, Wang C. Liposclerosing myxofibrous tumor of the distal femur: A case report. Frontiers in Surgery. 2023-01-06 2023;9doi:10.3389/fsurg.2022.1009975
- Beytemür O, Tetikkurt Ü S, Albay C, Kavşut G, Güleç A. Liposclerosing myxofibrous tumor: a rare tumor of proximal femur. Eklem Hastalik Cerrahisi. Dec 2017;28(3):210-3. doi:10.5606/ehc.2017.48394
- Deel C, Hassell L. Liposclerosing Myxofibrous Tumor: A Review. Archives of Pathology & Laboratory Medicine. 2016-05-01 2016;140(5):473-476. doi:10.5858/2014-0503-rs
- Bahk WJ, Seo KJ. Malignant transformation of liposclerosing myxofibrous tumour. Pathology. 2021;53(5):660-663. doi:10.1016/j.pathol.2020.09.032
- Regado ER, Garcia PB, Caruso AC, et al. Liposclerosing myxofibrous tumor: A series of 9 cases and review of the literature. J Orthop. Sep 2016;13(3):136-9. doi:10.1016/j.jor.2016.03.003
- Ploof J, Shaikh H, Melli J, Jour G, Turtz A. Liposclerosing Myxofibrous Tumor of the Cranial Vault: A Case Report. Neurosurgery. 2019;84(3):E207-E210. doi:10.1093/neuros/nyy071
- Campbell K, Wodajo F. Two-step Malignant Transformation of a Liposclerosing Myxofibrous Tumor of Bone. Clinical Orthopaedics & Related Research. 2008-11-01 2008;466(11):2873-2877. doi:10.1007/s11999-008-0362-9
- Dattilo J, McCarthy EF. Liposclerosing myxofibrous tumor (LSMFT), a study of 33 patients: should it be a distinct entity? Iowa Orthop J. 2012;32:35-9.
- Barnds B, Grote C, Mettman D, Templeton K. Liposclerosing Myxofibrous Tumor in a Patient with Prostate Cancer: A Case Report. JBJS case connector. 2019;9(4):e0411-e0411. doi:10.2106/JBJS.CC.18.00411
- Heim-Hall JM, Williams RP. Liposclerosing myxofibrous tumour: a traumatized variant of fibrous dysplasia? Report of four cases and review of the literature. Histopathology. Oct 2004;45(4):369-76. doi:10.1111/j.1365-2559.2004.01951.x
- Corsi A, De Maio F, Ippolito E, et al. Clinical Vignette: Monostotic Fibrous Dysplasia of the Proximal Femur and Liposclerosing Myxofibrous Tumor: Which One Is Which? Journal of Bone and Mineral Research. 2006-12-01 2006;21(12):1955-1958. doi:10.1359/jbmr.060818
- Moya-Angeler J, Gianakos AL, Villa JC, Ni A, Lane JM. Current concepts on osteonecrosis of the femoral head. World J Orthop. Sep 18 2015;6(8):590-601. doi:10.5312/wjo.v6.i8.590
- Konarski W, Poboży T, Śliwczyński A, et al. Avascular Necrosis of Femoral Head—Overview and Current State of the Art. International Journal of Environmental Research and Public Health. 2022-06-15 2022;19(12):7348. doi:10.3390/ijerph19127348
- Hines JT, Jo WL, Cui Q, et al. Osteonecrosis of the Femoral Head: an Updated Review of ARCO on Pathogenesis, Staging and Treatment. J Korean Med Sci. Jun 21 2021;36(24):e177. doi:10.3346/jkms.2021.36.e177
- Motta F, Timilsina S, Gershwin ME, Selmi C. Steroid-induced osteonecrosis. J Transl Autoimmun. 2022;5:100168. doi:10.1016/j.jtauto.2022.100168
- Yoon BH, Jones LC, Chen CH, et al. Etiologic Classification Criteria of ARCO on Femoral Head Osteonecrosis Part 2: Alcohol-Associated Osteonecrosis. J Arthroplasty. Jan 2019;34(1):169-174.e1. doi:10.1016/j.arth.2018.09.006


